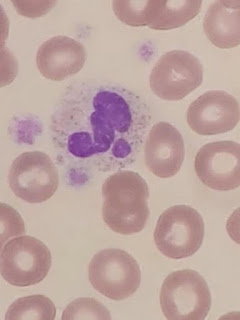
Zini et al, 2012
I recently reviewed the ICSH recommendations for counting schistocytes in a suspected Thrombotic Microangioapathy and have proposed we do this in our laboratory.
Proposal
To provide a percentage schistocyte count on those films
where there is a true thrombocytopenia in conjunction with schistocytes.
The ICSH gives normal reference value of <1 %
schistocytes in healthy adults and full term neonates and <5% in premature
neonates2 .It is suggested that schistocyte counts greater than
these values are a robust morphological threshold for suspecting red cell
mechanical damage due to thrombotic microangiopathy (TMA). A lack of
schistocytes does not rule out a TMA however.
When
not to perform a schistocyte count
Schistocytes are seen in a number of conditions and are not
specific to TMA. Schistocytes caused by a TMA are usually the dominant feature
on the blood film, perhaps in conjunction with moderate signs of stimulated
erythropoiesis such as polychromasia, basophilic stippling and nucleated red
cells.
If schistocytes are present alongside multiple other red
cell abnormalities, the percentage schistocyte count is not appropriate.
Criteria
for schistocyte recognition
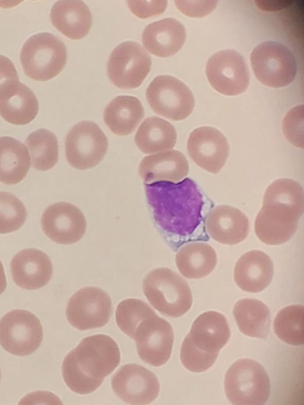
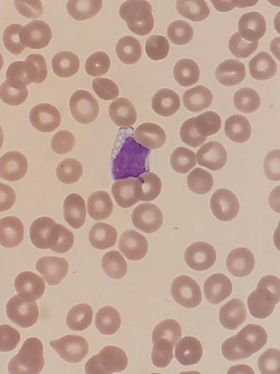
Schistocytes are defined by the ICSH as always smaller than
intact red cells and can have the shape of fragments with sharp angles and
straight borders, small crescents, helmet cells, keratocytes, or
microspherocytes. Microspherocytes only to be included in the presence of other
shapes mentioned1
Bite cells, spherocytes, irregularly contracted cells, tear
drop poikilocytes, and echinocytes should not be included in a schistocyte
count.
(See photograph above)
Method
Quantify the schistocytes, only if it they are the dominant
abnormality on the film. Not appropriate if they are present alongside multiple
other red cell abnormalities.
·
Schistocytes will be counted using a high power
x 100 objective.
·
Work out by counting, approximately how many
red cells are present per field On a well spread normal film this is
approximately 200.
·
In each field count how many schistocytes you
see until you have covered 1000 red cells.
· Express as a percentage of red cells.
Example
Worked out that there are approximately
200 red cells per high power field. Need to count how many fragments are in
10000 red cells. Therefore, five fields of 200 cells will give a total of 10000
red cells.
Field
1
200 red cells, 10
schistocytes seen
Field
2
200 red cells, 6 schistocytes seen
Field
3
200 red cells, 8 schistocytes seen
Field
4
200 red cells, 8 schistocytes seen
Field
5
200 red cells, 9 schistocytes seen
10+6+8+8+9 = 41 schistocytes seen in a
10000 red cells
41 ![]() 10000 x 100 = a schistocyte count of 4.1%
10000 x 100 = a schistocyte count of 4.1%
Schistocyte
recognition.
Zini et al, 2021
References
1 Zini
G, d'Onofrio G, Biggs C, et al. ICSH recommendations for
identification, diagnostic value, and quantitation of schistocytes. Int
J Lab Hematol. 2012; 34: 107-116.
2 Zini G, d'Onofrio G, Erber WN, Lee SH, Nagai Y, Basak GW,
Lesesve JF; International Council for Standardization in Hematology (ICSH).
2021 update of the 2012 ICSH Recommendations for identification, diagnostic
value, and quantitation of schistocytes: Impact and revisions. Int J Lab
Hematol. 2021 Dec;43(6):1264-1271. doi: 10.1111/ijlh.13682. Epub 2021 Aug 24.
PMID: 34431220.